Background: The dimorphism at position -21 of exon 1 in HLA-B gives rise to leader peptides featuring either methionine (M) or threonine (T) as the second residue in the processed leader peptide (HLA-B leader). The cell surface abundance of HLA-E is positively correlated with the copy number of −21M HLA-B. In hematopoietic cell transplantation (HCT), the MM genotype in the recipients is associated with worse outcomes after HLA-matched and mismatched unrelated bone marrow and peripheral blood stem cell transplantation compared with TT (Kanaya M, et al. ASH Annual Meeting 2021, Petersdorf EW et al. Lancet Haematol. 2020 and Blood. 2020). Eurocord/EBMT study revealed HLA-B leader mismatch in HLA-B mismatched combination is associated with relapse and non-relapse mortality (NRM) risks after cord blood transplantation (CBT) (Petersdorf EW et al. Haematologica 2021). Notably, approximately 70% of patients in the literature administrated anti-thymocyte globulin (ATG) for graft-versus-host disease (GVHD) prophylaxis. The impacts of HLA-B leader dimorphism on the outcomes of CBT still needed to be elucidated more. Particularly, we were interested in the significance of HLA-B leaders in CBT without an ATG, then we conducted a retrospective study to elucidate it.
Methods: Clinical data of 10,569 patients who underwent single-unit CBT for acute myeloid leukemia (AML, n=5,518), myelodysplastic syndrome (MDS, n=1,483), acute lymphoblastic leukemia (ALL, n=2,020), and lymphoma (n=1,548) between 2008 - 2021 were provided by the Japanese Data Center for Hematopoietic Cell Transplantation. HLA-A, -B, -C, and -DRB1 typing was performed at least at the antigen level. The leader genotype is defined based on the two HLA-B allotypes. All patients were not used ATG for GVHD prophylaxis. Study outcomes were overall survival (OS), cumulative incidences of relapse, NRM, and grade II-IV acute GVHD (aGVHD). Multivariable Cox regression analysis including covariables; disease risk index (DRI), myeloablative/reduced-intensity conditioning, presence of total body irradiation as a conditioning regimen, short-term methotrexate or mycophenolate mofetil for GVHD prophylaxis, recipient cytomegalovirus serostatus. All statistical analyses were performed with EZR.
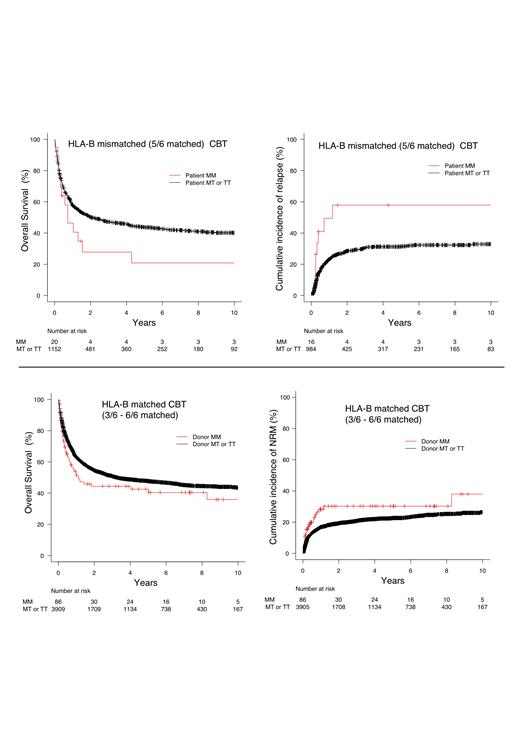
Results: The median patient's age was 53 (range 0-88) years. The numbers of patients with HLA-B leader TT, MT, and MM genotype were 7,639 (72.3 %), 2,707 (25.6 %), and 223 (2.1 %), respectively. To classify HLA-B match/mismatch status, we stratify “HLA-B one locus mismatched (GVH direction)” and HLA-B matched (no GVH direction mismatch)“. First, we focused on HLA-B one antigen mismatched (HLA 5/6 antigen matched, n = 1172) setting as shown in the previous literature. Recipient MM genotype showed significantly worse OS and relapse in the multivariate analysis compared to MT or TT genotype (Tx) (Figure) (OS: hazard ratio [HR] 0.543 [95% CI, 0.311 - 0.947]; p = 0.032, relapse: [HR] 2.863 [95% CI, 1.330 - 6.165]; p = 0.007). Recipient MM didn't relate to relapse and grade II-IV aGVHD.
Next, we explored the impacts of HLA-B leaders in HLA-B antigen-matched CBT (3/6 - 6/6 matched, n = 3995). Donor MM genotype demonstrated a negative impact for OS and NRM, not relapse and aGVHD, compared to Tx donor (Figure) (OS: [HR] 0.738 [95% CI, 0.542 - 1.000]; p 0.050, NRM: [HR] 1.695 [95% CI, 1.097 - 2.621]; p = 0.018). Especially, OS and NRM of donor MM genotype were significantly inferior to Tx in HLA 6/6 matched (n = 914) CBT (OS: [HR] 0.311 [95% CI, 0.187 - 0.518]; p < 0.001, NRM: [HR] 3.216 [95% CI, 1.931 - 5.356]; p = 0.034).
Conclusions: ATG would affect immune dynamics strongly in CBT via in-vivo T cell depletion, yet the current study demonstrated the HLA-B leader was still a significant risk factor in CBT without ATG for the first time. Recipient MM genotype in HLA-B one locus mismatched (HLA 5/6 matched) and donor MM genotype in HLA-B matched were the risk factors for worse CBT outcomes. We concluded MM genotype donors would be avoided to decrease NRM in HLA-B matched CBT. The precise mechanism of negative impact in CBT outcomes in patients or donor MM genotype remains to be clarified, however, we assume higher level HLA-E expression may be crucial in terms of NKG2A inhibitory signaling to T cells/NK cells and T-cell direct recognition for peptide/HLA-E complex in CBT.
Disclosures
Kanaya:Meiji Seika Pharma Co., Ltd: Honoraria; Sanofi K.K.: Honoraria; JANSSEN PHARMACEUTICAL K.K.: Honoraria; Chugai Pharmaceutical Co., Ltd. l: Honoraria; Astellas Pharma Inc.: Honoraria. Hashimoto:Daiichi Sankyo Inc: Honoraria; Ono Pharma: Honoraria; Janssen Pharma: Honoraria; Kyowa-Kirin: Honoraria; Chugai Pharmaceutical: Honoraria; LUCA Science: Patents & Royalties; Astellas Pharma: Honoraria. Tanaka:Astellas Phrama: Speakers Bureau; Chugai Pharmaceutical: Speakers Bureau; Kyowa-Kirin: Speakers Bureau; Daiichi Sankyo: Speakers Bureau; MSD: Speakers Bureau; Otsuka Pharmaceutical: Speakers Bureau; Pfizer: Speakers Bureau; Sumitomo Pharma: Speakers Bureau; Asahi Kasei Pharma: Speakers Bureau; Abbvie: Speakers Bureau. Takahashi:Daiichi Sankyo RD Novare, Otsuka Pharmaceutical: Research Funding; Nippon Sinyaku: Honoraria; Novartis: Honoraria; Chugai Pharmaceutical: Honoraria; Asahi Kasei Pharma: Honoraria; AstraZeneca: Honoraria; Terumo: Honoraria; Kyowa Kirin: Honoraria; JCR pharma: Honoraria. Sawa:Sanofi: Honoraria; Janssen: Honoraria. Ichinohe:Repertoire Genesis: Research Funding. Atsuta:JCR Pharmaceuticals Co., Ltd.: Consultancy; Novartis Pharma KK: Speakers Bureau; Meiji Seika Pharma Co, Ltd.: Honoraria; CHUGAI PHARMACEUTICAL CO., LTD.: Speakers Bureau; Otsuka Pharmaceutical Co., Ltd: Speakers Bureau. Morishima:Chugai Pharma: Honoraria; Abbvie: Honoraria; Kyowa Kirin: Honoraria; Pfizer: Honoraria; Daiichi Sankyo: Honoraria; Sanofi: Honoraria; Janssen: Honoraria.